From pv magazine Global
Sodium-ion batteries offer considerable advantages in terms of raw material costs and safety, but manufacturing costs need to fall further in order for them to compete on par with the ubiquitous lithium-ion chemistry, particularly in stationary and large-scale storage applications.
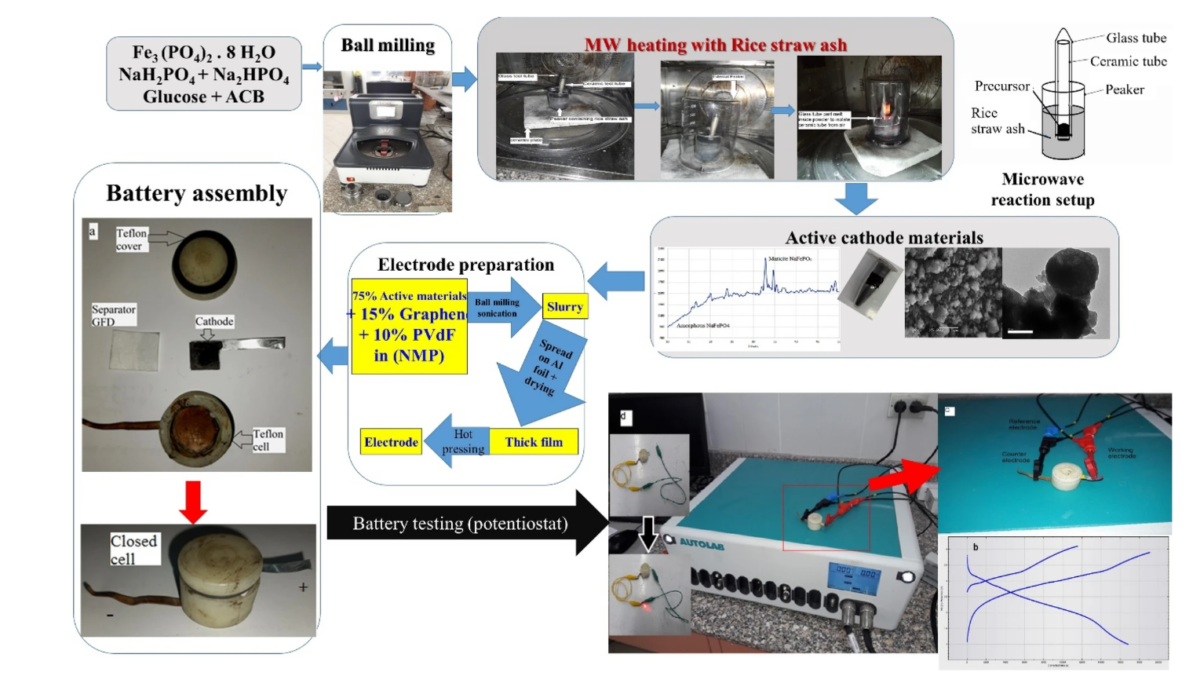
To drive down cell prices as much as possible, researchers at City of Scientific Research and Technological Applications in Alexandria, Egypt, have developed a new synthesis method for sodium-ion battery cathodes that greatly reduces the cost and time needed for each preparation step.
Using Egyptian rice straw ash as a microwave absorber, the researchers synthesised, crystallised, and carbon-coated mixtures of sodium iron phosphates-carbon composites (NaFePO4-C).
The sodium ion electroactive composites were prepared using different microwave heating durations, ranging from 30 to 100 seconds. According to the researchers, the optimum synthesis time was just 60 seconds for applications in sodium-ion batteries, and as a supercapacitor.
They said that high-purity inert gases were not needed during synthesis, processing, or cell assembly, resulting in lower costs.
“The time and cost of energy consumed for the synthesis are incomparable,” said the researchers in “Ultra-fast green microwave assisted synthesis of NaFePO4 -C nanocomposites for sodium ion batteries and supercapacitors” which was recently published in Nature.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.








By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.