A team of researchers from Monash University’s Faculty of Engineering have developed a new lithium-sulphur (Li-S) battery design featuring a nanoporous polymer-coated lithium foil anode that “significantly” improves the number of times the battery can be cycled.
Lead researcher Declan McNamara said when compared with similar coated lithium anode systems, the polymer coating cell has exhibited outstanding performance across a range of metrics.
“This coating is a step towards highly efficient, easily manufactured Li-S batteries,” he said.
Li-S battery technology continues to prove popular among researchers and commercial developers, with the potential for the metallic lithium and sulphur combination to deliver more energy per gram than lithium-ion batteries.
The technology does however have its limitations. Typically, Li-S batteries contain a lithium anode (negative electrode) and sulphur cathode (positive electrode) with a separating layer. When the battery charges and discharges, the lithium and sulphur react which leads to the formation of polysulfides, rapidly reducing the battery performance.
“Metallic lithium is a bit of a double-edged sword,” McNamara said. “Lithium is packed full of energy, but in a bad battery, this energy is wasted on side reactions. On the other hand, if the energy is channelled correctly, it can make some incredible energy storage devices that are easier to make.”
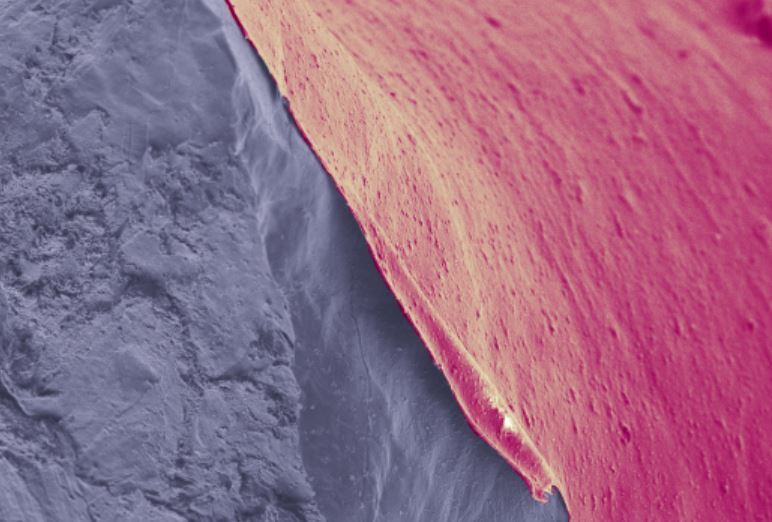
In this new battery configuration, the researchers at Monash have coated a lithium foil anode with a nanoporous polymer which they say allows the lithium ions to move through while protecting the anode from corrosive sulphur compounds.
“The polymer contains tiny holes less than a nanometre in size, one billionth of a metre, which allow lithium ions to move freely while blocking other chemicals that would attack the lithium,” McNamara said. “The coating also acts as a scaffold for lithium, and helps it charge and discharge repeatedly.”
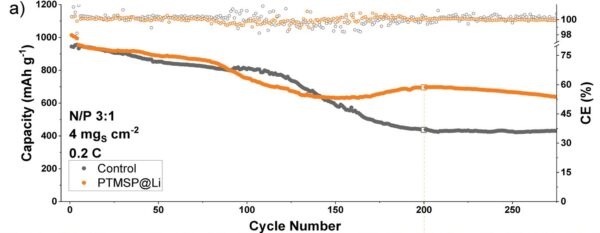
Image: Monash University
In their paper, the researchers say the polymer coated anode had exhibited improved capacity retention over 275 cycles, adding that the new design reduces the amount of lithium required in a single battery and does not require nickel or cobalt, removing the need for minerals that have a significant environmental and social cost.
Mainak Majumder, from Monash’s Faculty of Engineering, said these developments are promising steps towards more widespread adoption of Li-S batteries and other lithium metal-based energy storage systems.
“The study establishes a new framework to protect Li-metal from rapid decay or catastrophic failure which has been an Achilles heel for Li-S batteries,” he said.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.








By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.