A research team from the University of New South Wales (UNSW) say they have developed a technique for sustainable ammonia production at scale. Technically dense, the method reportedly does not require the high temperatures, high pressure and big infrastructure currently needed to produce ammonia – a compound mostly used in agricultural fertilisers, but which has also become crucial for hydrogen proponents.
The technique builds on previous research from the same UNSW team, with lead researcher Dr. Ali Jalili explaining then that his group and colleagues had devised proof-of-concept lab experiments which used plasma, a form of lightning made in a tube, to convert atmospheric nitrogen in air into an intermediary known as NOx – either NO2- (nitrite) or NO3- (nitrate). The nitrogen in these compounds is much more reactive than N2 in the air.
“Working with our University of Sydney colleagues, we designed a range of scalable plasma reactors that could generate the NOx intermediary at a significant rate and high energy efficiency,” he said. “Once we generated that intermediary in water, designing a selective catalyst and scaling the system became significantly easier. The breakthrough of our technology was in the design of the high-performance plasma reactors coupled with electrochemistry.”
The foundation of this research has been licensed to an Australian industry partner, PlasmaLeap Technologies, but the team have now published a new paper in the journal Applied Catalysis B: Environmental. In it, they say they have made significant advances in energy efficiency and production rate, thus improving commercial profitability.
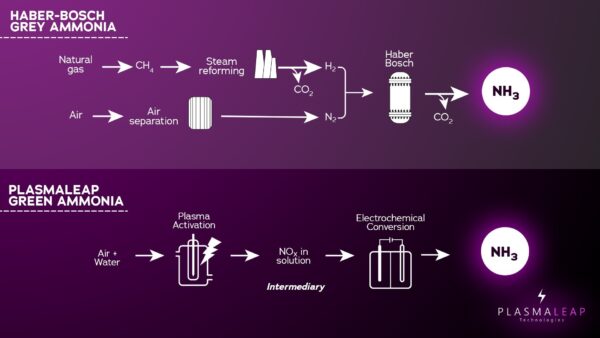
Image: PlasmaLeap
In this new study, the team said they “explored the integration of non-thermal plasma and electrochemical processes.”
“However, the success of this hybrid process is contingent on the energy efficiency of the plasma-generated species. Herein, we developed a plasma bubble reactor, driven by nanosecond pulses interfacing plasma directly with water.”
“By using on-and-off plasma ignition with rapid pulses, we could regulate energy consumption in cycles and achieved the lowest reported energy consumption of 2.7 ± 0.1 kWh mol-1 NO3− and 3.2 ± 0.1 kWh mol-1 NH4+ after electrocatalytic nitrate reduction.”
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.









By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.